Please use a PC Browser to access Register-Tadawul
Ultragenyx BLA Resubmission Puts UX111 And RARE Valuation In Focus
Ultragenyx Pharmaceutical, Inc. RARE | 21.25 | -0.98% |
- Ultragenyx Pharmaceutical resubmitted its Biologics License Application to the FDA for UX111, a gene therapy candidate for Sanfilippo syndrome type A.
- The filing includes long term clinical data and responses to earlier regulatory concerns about the therapy.
- If accepted, UX111 could become the first approved treatment specifically targeting this rare and serious condition.
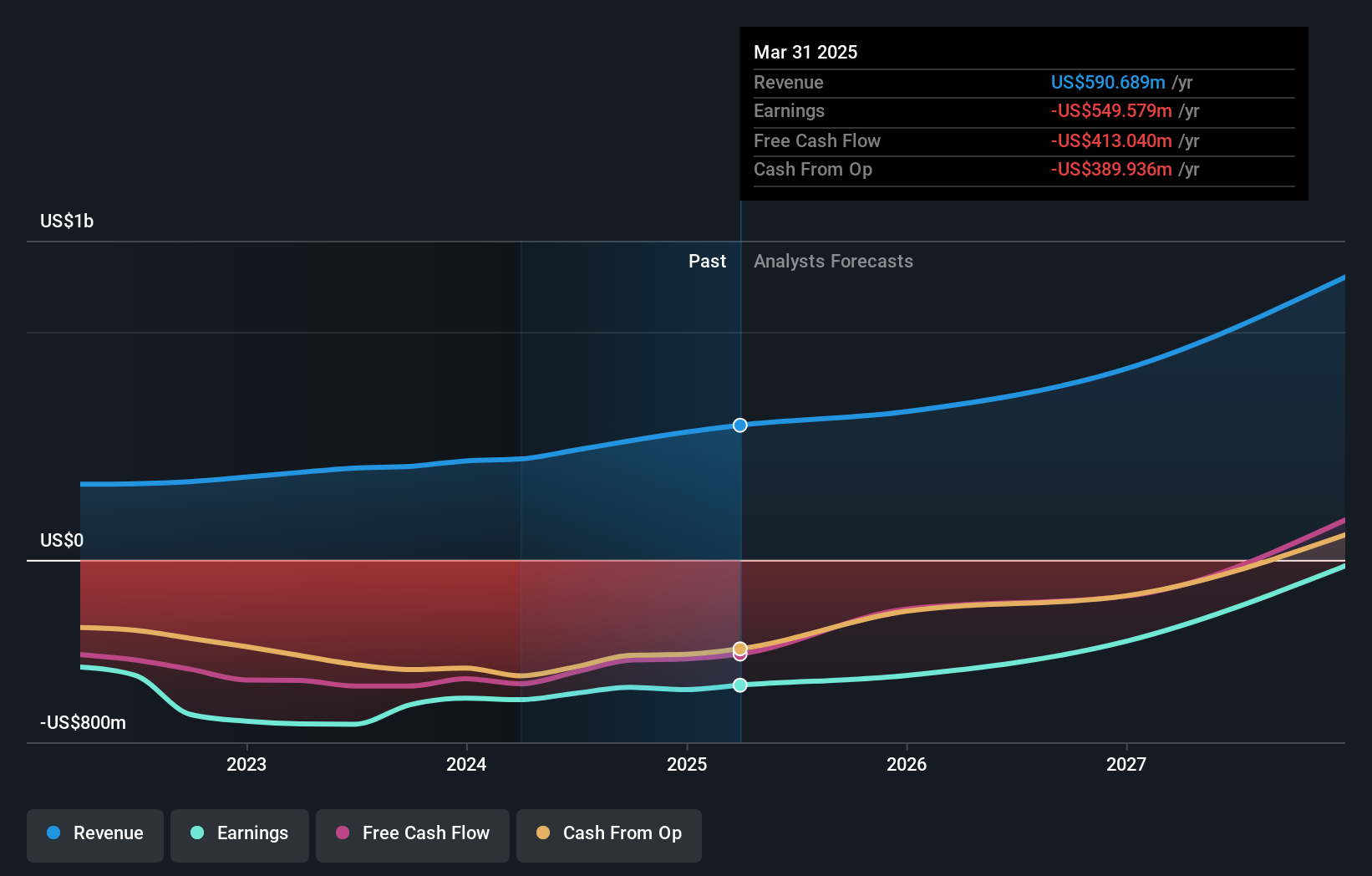
Ultragenyx Pharmaceutical (NasdaqGS:RARE), trading at $24.07 as of the last close, is moving ahead with a key regulatory step for its rare disease pipeline. The stock has seen a 44.1% decline over the past year and a 47.4% decline over three years, reflecting the risks and uncertainty that can come with a focus on complex gene therapies.
The UX111 resubmission puts fresh attention on how regulators assess gene therapies for ultra rare conditions and how Ultragenyx manages scientific and regulatory hurdles. For investors, the next updates from the FDA process, along with any additional clinical disclosures, will be important markers for understanding how this program could influence the overall story for NasdaqGS:RARE.
Stay updated on the most important news stories for Ultragenyx Pharmaceutical by adding it to your watchlist or portfolio. Alternatively, explore our Community to discover new perspectives on Ultragenyx Pharmaceutical.
Quick Assessment
- ✅ Price vs Analyst Target: At US$24.07 versus a US$64.00 analyst target, the price sits about 62% below consensus.
- ✅ Simply Wall St Valuation: The shares are described as trading 92.7% below the estimated fair value, flagging a large valuation gap.
- ✅ Recent Momentum: A 30 day return of roughly 2.0% shows modest positive short term momentum into the resubmission.
Check out Simply Wall St's in depth valuation analysis for Ultragenyx Pharmaceutical.
Key Considerations
- 📊 The FDA resubmission, backed by long term UX111 data, keeps a potential first in disease gene therapy in play for the Ultragenyx story.
- 📊 Watch for FDA acceptance milestones, labeling questions and any new safety or efficacy data that could shift how UX111 is viewed in the portfolio.
- ⚠️ With two identified risk flags including a short cash runway and share price volatility, funding needs and dilution risk stay front of mind if timelines extend.
Dig Deeper
For the full picture including more risks and rewards, check out the complete Ultragenyx Pharmaceutical analysis.
This article by Simply Wall St is general in nature. We provide commentary based on historical data and analyst forecasts only using an unbiased methodology and our articles are not intended to be financial advice. It does not constitute a recommendation to buy or sell any stock, and does not take account of your objectives, or your financial situation. We aim to bring you long-term focused analysis driven by fundamental data. Note that our analysis may not factor in the latest price-sensitive company announcements or qualitative material. Simply Wall St has no position in any stocks mentioned.



