Ascendis Pharma Achondroplasia Data Highlights Emerging Rare Disease Franchise Potential
Ascendis Pharma A/S Sponsored ADR ASND | 229.25 | +0.44% |
- Ascendis Pharma (NasdaqGS:ASND) reported positive topline Week 52 results from its COACH Phase 2 trial in children with achondroplasia.
- The combination therapy showed durable growth, improved body proportionality, and a favorable safety profile over the 52 week period.
- The data support the company’s achondroplasia program as a potential backbone treatment approach for pediatric patients.
Ascendis Pharma, a biopharmaceutical company focused on endocrinology related rare diseases, is sharpening investor attention with this new set of Phase 2 data. For readers following the achondroplasia space, the COACH trial adds fresh clinical information on how combination regimens might address growth and proportionality in children who currently have limited treatment choices.
For you as an investor, the key questions now center on how these Phase 2 outcomes feed into later stage trials, regulatory discussions, and eventual real world use if approved. The COACH readout may help shape how Ascendis positions its investigational therapies within the broader treatment toolkit for pediatric achondroplasia over time.
Stay updated on the most important news stories for Ascendis Pharma by adding it to your watchlist or portfolio. Alternatively, explore our Community to discover new perspectives on Ascendis Pharma.
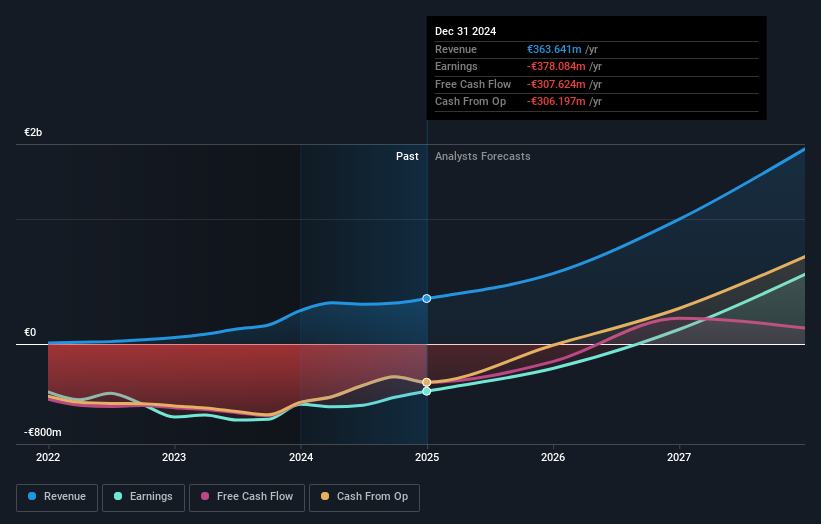
The COACH Phase 2 data points to a broader product story for Ascendis Pharma in achondroplasia, because the combination of TransCon CNP and TransCon hGH is linked not only to higher annualized growth velocity but also to body proportionality and arm span, with safety described as in line with each monotherapy. For you, that speaks to a potential multi asset franchise, where TransCon CNP under Priority Review could serve as a core therapy and TransCon hGH, already marketed as SKYTROFA in growth hormone deficiency, might extend its role into a new rare disease segment if future studies and regulators support that path.
How This Fits Into The Ascendis Pharma Narrative
The existing Ascendis narrative already centers on TransCon based therapies and the idea that platform based products across Yorvipath, SKYTROFA and TransCon CNP can reduce reliance on any single drug. These COACH results align with that storyline by adding clinical support for combination use in achondroplasia, which may influence how some investors think about the depth of the rare disease opportunity alongside ongoing launches and label expansion efforts highlighted in analyst commentary.
Risks and Rewards To Keep In Mind
- Combination data in children with achondroplasia support the view that TransCon CNP could act as a backbone therapy with TransCon hGH providing added benefit, which broadens the potential use case within the rare endocrine portfolio.
- All children completing 52 weeks of treatment with generally mild treatment emergent adverse events and a safety profile described as consistent with monotherapies may reassure some investors about tolerability in a pediatric setting.
- Ascendis still faces execution risk around regulatory review timelines, label outcomes and future payer decisions, and analysts have flagged at least one key risk for the company overall.
- Competition in achondroplasia and rare endocrinology, together with ongoing R&D and commercial spending needs, could affect how quickly any achondroplasia revenue stream contributes relative to expectations.
What To Watch Next
From here, you may want to track the FDA Priority Review process for TransCon CNP, further COACH updates and how analysts adjust their achondroplasia assumptions alongside real world trends for SKYTROFA and Yorvipath, and you can stay plugged into evolving views by following the latest Community Narratives on Simply Wall St.
This article by Simply Wall St is general in nature. We provide commentary based on historical data and analyst forecasts only using an unbiased methodology and our articles are not intended to be financial advice. It does not constitute a recommendation to buy or sell any stock, and does not take account of your objectives, or your financial situation. We aim to bring you long-term focused analysis driven by fundamental data. Note that our analysis may not factor in the latest price-sensitive company announcements or qualitative material. Simply Wall St has no position in any stocks mentioned.
