Merck’s New Ovarian Cancer Win And Women’s Health Push Tested By Valuation
Merck & Co., Inc. MRK | 120.87 | +0.02% |
- Merck (NYSE:MRK) received FDA approval for KEYTRUDA and subcutaneous KEYTRUDA QLEX for platinum resistant ovarian cancer, including a companion diagnostic.
- The approvals represent the first PD 1 inhibitors cleared for this patient group and introduce a less invasive subcutaneous option.
- Merck also entered an industry partnership to develop an intravaginal drug delivery platform, expanding its focus in women's health.
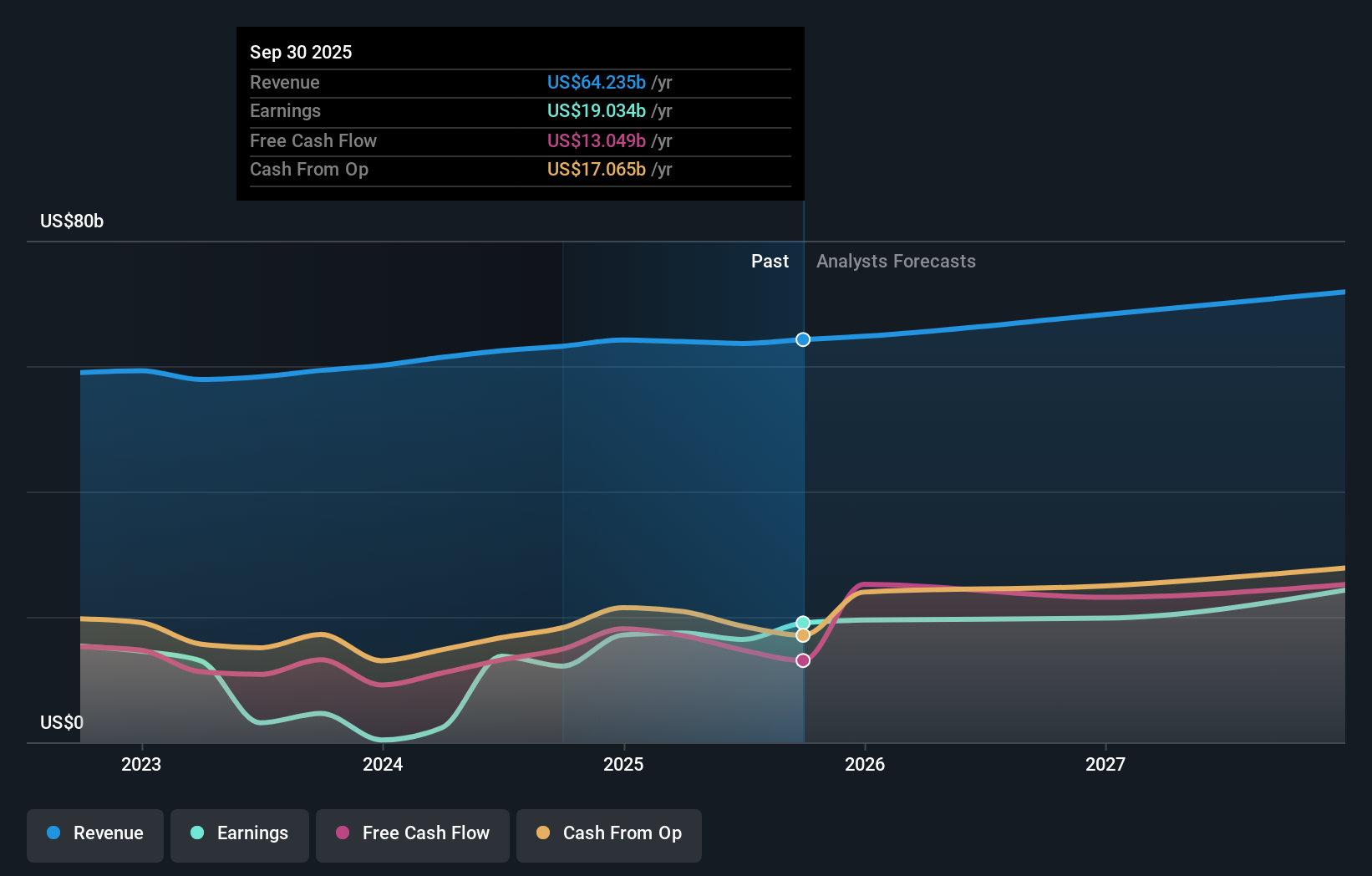
For investors watching NYSE:MRK, these developments arrive with the stock at $119.24 and a 1 year return of 46.6%. The company has also posted gains of 12.0% year to date and 94.4% over 5 years, which helps frame how the market has treated Merck's broader pipeline and execution.
Looking ahead, readers may want to track how uptake of the new ovarian cancer indication and subcutaneous formulation evolves, as well as how quickly the companion diagnostic is integrated into clinical practice. The new women's health alliance around intravaginal delivery could be another area to follow, particularly if it results in additional products or partnerships connected to Merck's portfolio.
Stay updated on the most important news stories for Merck by adding it to your watchlist or portfolio. Alternatively, explore our Community to discover new perspectives on Merck.
Quick Assessment
- ⚖️ Price vs Analyst Target: At US$119.24, Merck trades about 3.5% below the US$123.54 analyst consensus target, suggesting the price is close to where analysts see fair value.
- ✅ Simply Wall St Valuation: Simply Wall St currently views Merck as undervalued, trading 43.2% below its estimated fair value.
- ✅ Recent Momentum: The 30 day return of 10.1% shows positive short term momentum as the market reacts to developments such as the new KEYTRUDA approval and women's health partnership.
There is only one way to know the right time to buy, sell or hold Merck. Head to Simply Wall St's company report for the latest analysis of Merck's Fair Value..
Key Considerations
- 📊 The first in class PD 1 approval in platinum resistant ovarian cancer, together with a subcutaneous option, could reinforce KEYTRUDA's role in oncology and broaden the addressable patient pool.
- 📊 Watch uptake of the new indication, adoption of the companion diagnostic, and any product candidates that emerge from the intravaginal delivery partnership in women's health.
- ⚠️ Regulatory and execution risk remains, including real world safety and efficacy data, pricing pressure in oncology, and the need to manage any balance sheet concerns flagged in risk assessments.
Dig Deeper
For the full picture including more risks and rewards, check out the complete Merck analysis. Alternatively, you can visit the community page for Merck to see how other investors believe this latest news will impact the company's narrative.
This article by Simply Wall St is general in nature. We provide commentary based on historical data and analyst forecasts only using an unbiased methodology and our articles are not intended to be financial advice. It does not constitute a recommendation to buy or sell any stock, and does not take account of your objectives, or your financial situation. We aim to bring you long-term focused analysis driven by fundamental data. Note that our analysis may not factor in the latest price-sensitive company announcements or qualitative material. Simply Wall St has no position in any stocks mentioned.
