Sarepta’s SRP-1005 Trial Approval Opens New Huntington’s Disease Opportunity
Sarepta Therapeutics, Inc. SRPT | 22.80 | -1.85% |
- Sarepta Therapeutics (NasdaqGS:SRPT) received Medsafe approval in New Zealand for its first-in-human clinical trial of SRP-1005.
- SRP-1005 is an investigational siRNA therapeutic targeting Huntington's Disease, a severe neurodegenerative condition.
- The authorization allows Sarepta to begin a Phase 1 study, marking its clinical entry into Huntington's Disease research.
Sarepta Therapeutics is best known for its Duchenne muscular dystrophy franchise, and SRP-1005 broadens its rare disease focus into Huntington's Disease. For investors, the move into siRNA for another serious neurologic condition highlights how the company is applying its genetic medicine expertise in a new indication where disease-modifying options are limited.
Early stage trials such as this are primarily about safety and dosing, so it will take time before any efficacy picture becomes clearer. The start of human testing creates a new clinical catalyst track for NasdaqGS:SRPT that sits alongside its established Duchenne muscular dystrophy programs and may attract attention from investors following central nervous system drug development.
Stay updated on the most important news stories for Sarepta Therapeutics by adding it to your watchlist or portfolio. Alternatively, explore our Community to discover new perspectives on Sarepta Therapeutics.
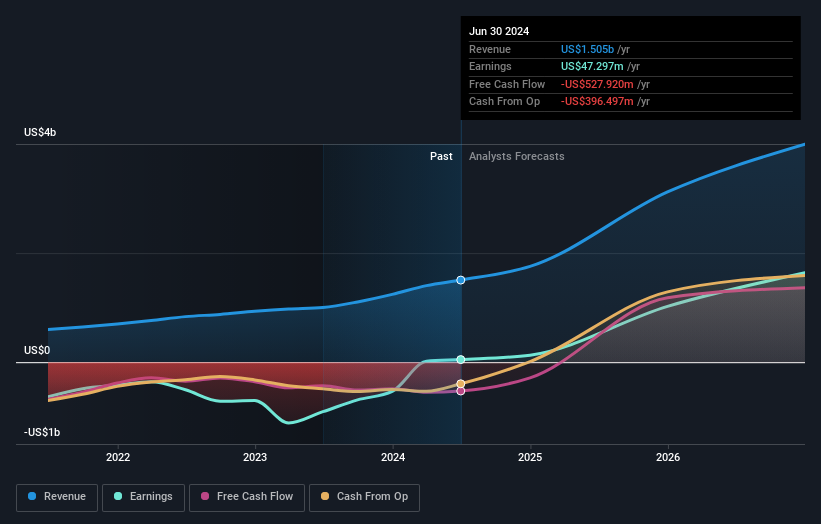
The Medsafe approval for INSIGHTT moves SRP-1005 from concept into human testing, which matters because it opens a new potential revenue stream outside Sarepta Therapeutics' core Duchenne muscular dystrophy franchise. Huntington's Disease is a rare, inherited condition with no approved treatments that address the root genetic cause, so any therapy that can safely lower the disease-driving protein could address a clear unmet need. A 24 patient, dose escalation Phase 1 trial is small by design and focused on safety and tolerability, so investors are unlikely to get near term clarity on commercial potential, but it does start to build clinical proof around Sarepta's siRNA platform. In a space where companies such as Ionis Pharmaceuticals and uniQure are also active, showing clean early safety data and a practical subcutaneous dosing profile may matter for differentiation later on. For Sarepta, progress in SRP-1005 could help balance sentiment that has been concentrated on gene therapy headlines and reinforces that the pipeline now spans multiple genetic medicine modalities rather than a single product story.
How This Fits Into The Sarepta Therapeutics Narrative
- This first in human trial in Huntington's Disease aligns with the narrative that expanding into siRNA programs can diversify Sarepta's pipeline and add new potential revenue streams beyond gene therapy for Duchenne muscular dystrophy.
- Executing another early stage program could also stretch research and development resources and add to already high research costs that the narrative highlights as a pressure point for profitability.
- The narrative focuses heavily on ELEVIDYS and limb girdle muscular dystrophy programs, so the competitive and commercial implications of SRP-1005 in Huntington's Disease may not yet be fully reflected in how investors frame Sarepta's long term story.
Knowing what a company is worth starts with understanding its story. Check out one of the top narratives in the Simply Wall St Community for Sarepta Therapeutics to help decide what it's worth to you.
The Risks and Rewards Investors Should Consider
- ⚠️ Early stage trials carry meaningful development risk, and any safety concerns in this small first in human study could affect confidence in both SRP-1005 and Sarepta's broader siRNA platform.
- ⚠️ Building out another complex rare disease program increases funding needs and execution complexity, which could weigh on margins if trial timelines extend or additional studies are required.
- 🎁 Entering Huntington's Disease with a genetic medicine that targets the underlying cause gives Sarepta exposure to a new patient population where existing options are limited.
- 🎁 Progress in SRP-1005 adds to a broader pipeline that already includes gene therapy and other RNA targeted programs, which can help reduce reliance on a single product and create more balanced long term revenue potential.
What To Watch Going Forward
From here, the key milestones to follow are trial initiation timing in the second quarter of 2026, enrollment progress for the roughly 24 planned participants, and any early safety or dosing updates Sarepta shares from INSIGHTT. Investors may also want to track how management discusses SRP-1005 on upcoming earnings calls relative to its gene therapy portfolio, since that can signal how central Huntington's Disease is becoming to the overall growth plan. Competitive developments from other Huntington's Disease players such as Ionis Pharmaceuticals and uniQure will help frame how differentiated a subcutaneous siRNA approach could be if it advances. Finally, watch for any commentary on research spending tied to siRNA programs, as that will influence how quickly Sarepta aims to move SRP-1005 through later stage trials if early data are supportive.
To ensure you're always in the loop on how the latest news impacts the investment narrative for Sarepta Therapeutics, head to the community page for Sarepta Therapeutics to never miss an update on the top community narratives.
This article by Simply Wall St is general in nature. We provide commentary based on historical data and analyst forecasts only using an unbiased methodology and our articles are not intended to be financial advice. It does not constitute a recommendation to buy or sell any stock, and does not take account of your objectives, or your financial situation. We aim to bring you long-term focused analysis driven by fundamental data. Note that our analysis may not factor in the latest price-sensitive company announcements or qualitative material. Simply Wall St has no position in any stocks mentioned.
